The Hydrogen Engine
The Properties of Hydrogen Fuel
Hydrogen is available in abundance and is the most naturally occurring fuel in nature. It is present in water, in acids, in all vegetation and animal protein. Coal and petroleum contain it and the distant sun and stars contain large quantities of hydrogen.
 |
Hydrogen is a gaseous fuel and since it always leaves behind a little water after burning, it was named hydrogen (from a Greek word meaning “water-maker”). Being the lightest gas known to man, there is scarcely any hydrogen in atmospheric air as it would just float away, out of the earth’s atmosphere.
The distinct difference in obtaining hydrogen is that it cannot be mined or harvested like petroleum. There are no oil rigs to build and no hydrogen fields to mine. Instead, hydrogen has to be extracted from its naturally occurring source. If you take two wires and dip it into a jar of water and run electricity through it, bubbles of gas appear at the ends of the wires. The positive pole (anode) gives off oxygen while the negative pole (cathode) gives off hydrogen. Theoretically, all the water will be transformed into the gases hydrogen and oxygen until there is no more water left in the jar. This process is called electrolysis. This is the simplest method of obtaining hydrogen, but it is far from efficient. The amount of energy it takes to split water molecules into hydrogen and oxygen is much more than the amount of energy it produces. Hydrogen can also be produced by dipping metals into an acidic solution, or extracted from hydrocarbon fuels but these methods leave behind residues which are potential pollutants.
The combustive properties of hydrogen makes it the richest and most efficient energy source known to man, it produces no pollutants, is abundantly available and completely renewable as a fuel source. Liquid hydrogen has the best energy-to-weight ratio, which has made it the fuel of choice for today’s rocket engines. Harnessing the power of the hydrogen has become one of today’s most highly sought after technological breakthroughs.
Among the properties of the hydrogen which contributes to its use as a fuel source :-
- Wide flammability range.
Compared to other fuels, hydrogen is not so fussy with its density mixture. It can ignite anywhere from a Fuel-to-Air mixture of 4 to 74 percent. - Easily ignitable.
Hydrogen ignites easier than gasoline. This provides for efficient and prompt ignition but the drawback is hot spots within the combustion chamber can cause premature ignition. - High expansion mass.
The expanding gases formed by spent hydrogen has a much higher velocity and mass compared to gasoline. - Easily dispersed.
The ability of hydrogen to blend with air is greater than gasoline, thus forming a more uniform mixture. - Low density.
Hydrogen occupies a very large volumetric area in its gaseous state. To facilitate the storage of hydrogen gas, it has to be stored in its liquid form within high pressure tanks. - Low boiling temperature.
Liquid hydrogen cannot be mixed with other liquid fuels. Its low boiling point (-252 C) will freeze other fuels. This means a separate storage tank is needed to store liquid hydrogen.
Some theories abound that hydrogen is a leading contender to replace fossil fuels in the not so distant future. With the rapid advancement of hydrogen fuel cell technology, electrically powered cars might become practical enough to eventually replace internal combustion engines. The advantage is that the combustion of hydrogen produces only water, whereas fossil fuels produces carbon dioxide and carbon monoxide, a pollutant contributing to global warming.
However, solutions will have to be found in the area of producing hydrogen. The current most widely used method of electrolysis consumes more energy than it produces. Other simpler methods include extracting hydrogen from fossil fuels, but this method produces pollutants in the process, defeating the purpose of a zero emissions fuel source. A relatively new technology has been developed to produce hydrogen on demand by splitting water molecules, but this has other challenges, namely the raw materials used is relatively precious and the scarcity of it is not enough for any practical use.
Finally, the issue of storage also has to be addressed. Liquid hydrogen has an extremely low density, 14 times lower than water and requires extensive insulation from high pressure tanks. The transfer of hydrogen between locations may also require pipes to be laid.
The Hydrogen Engine
There are two ways for automobile engines to utilize hydrogen fuel. The first is a Hydrogen Internal Combustion Engine or HICE. With HICE, hydrogen is injected directly into the combustion chamber and burnt as part of the fuel mixture. With slight modifications, many existing gasoline engines can be converted to run directly on hydrogen.
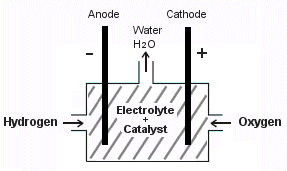
Another method of using hydrogen is through a fuel cell. With this method, hydrogen and oxygen is injected into a chamber where it reacts with a catalyst to produce electricity. They are very efficient and small fuel cells produce enough electricity to power electric cars. However fuel cells are very expensive to build.
 |
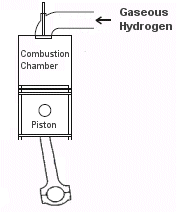 |
| A Hydrogen Fuel Cell produces electricity to power electric cars. | An Internal Combustion Hydrogen Engine burns hydrogen directly. |
Hydrogen Powered Engines - Development History
The first inventor of the hydrogen engine was Reverend W. Cecil James in 1820. His engine worked in a similar fashion as the steam engine, where he had used hydrogen to drive a piston. Although his design ran satisfactorily with a recorded revolution of 60 cycles per minute, but the explosions caused considerable noise. Forty years later, by 1860, many other gas engines had been developed by other inventors. Some of them had strange designs including double-acting versions where fuel was combusted on both sides of the piston. However none of them had any real commercial success with the main drawback being the excessive gas consumption.
With the discovery of gasoline in the 1860s and the invention of the carburetor in 1893, brought with it a new breakthrough in engineering. Gasoline engines could now be used both practically and safely, causing interest in other fuels to subside.
It was not until the 20th century that interest in hydrogen was again rekindled. It had found many uses including hydrogen filled airships, the hydrogen bomb, experimental hydrogen powered aircraft, and used most extensively in the rocket space program as rocket fuel.
In recent years, however, interest in hydrogen energy peaked at a feverish rate as concern for cleaner air and global warming fueled the search for an alternative energy source to replace fossil fuels. Various car manufacturers took to the challenge and began research and development of hydrogen powered cars.
The early 1990s saw the arrival of the Shelby Cobra, a freelance vehicle conversion project from gasoline to hydrogen power. Other more renowned car manufacturers such as Honda introduced their first prototype, the Honda FCX in 1999. A few years later in 2002, Mercedes Benz introduced their first generation of fuel cell vehicles. By 2004, Peugeot had joined this elite group and at the same time BMW’s Hydrogen Powered program produced the first breed of racecars to run on liquid hydrogen.
 |
 |
| A 1965 Ford Shelby Cobra | Hydrogen Powered BMW H2R |
Today, hydrogen powered prototypes and concept cars are being developed by Honda, Toyota, Nissan, Volkswagen, Hyundai, Audi, BMW, Mercedes Benz, Fiat, General Motors, Ford, Peugeot, Chrysler and other less prominent manufacturers. Many are approaching commercial viability such as Honda to begin limited marketing of their latest fuel cell vehicle in 2008. General Motors has stated that it can produce a commercially viable model by 2010.
|
We apologize but this website is still being updated with more content about Hydroxene and Hybrid Cars. Some or most of the links here do not work yet, but they are being completed as quickly as time permits.
If you encounter a broken link, then please re-visit us again about a week later. The last update is on December 29, 2006. When the website is complete, this message will be removed. |
